Key Takeaways
- The Periodic Table Chemistry Study Notes helps organize elements by atomic number and is essential for understanding properties and trends.
- The modern periodic table has 118 elements organized into 7 periods and 18 groups, showing specific similarities within groups.
- Elements are divided into blocks (s, p, d, f) based on electron configuration, influencing their chemical behavior.
- Key periodic trends include variations in atomic size, electronegativity, metallic nature, and ionization energy across periods and groups.
- Understanding these trends and the structure of the periodic table is vital for exams like SSC CGL 2026.
The periodic table is a chart that organizes all chemical elements based on their atomic number. It helps in understanding element properties, trends, and classifications. These notes are useful for SSC CGL 2026 and other exams like SSC CHSL, SSC MTS, and RRB NTPC.
What is the Periodic Table?
The periodic table is a systematic arrangement of elements in order of increasing atomic number. It is based on the Modern Periodic Law, which states that the properties of elements are periodic functions of their atomic numbers. Earlier, elements were arranged by atomic mass (Mendeleev), but modern classification uses atomic number.
Overview of the Modern Periodic Table
The modern periodic table contains 118 elements. In the modern periodic table, the elements are arranged according to their atomic numbers. The periodic table is divided into groups and periods. The vertical rows are called groups and horizontal rows are called periods. For the SSC CGL Exam, understanding the modern periodic table is essential as it forms the basis of many Chemistry questions in the General Awareness section.
| Feature | Details |
| Total Elements | 118 |
| Basis | Atomic Number |
| Periods | 7 (Horizontal rows) |
| Groups | 18 (Vertical columns) |
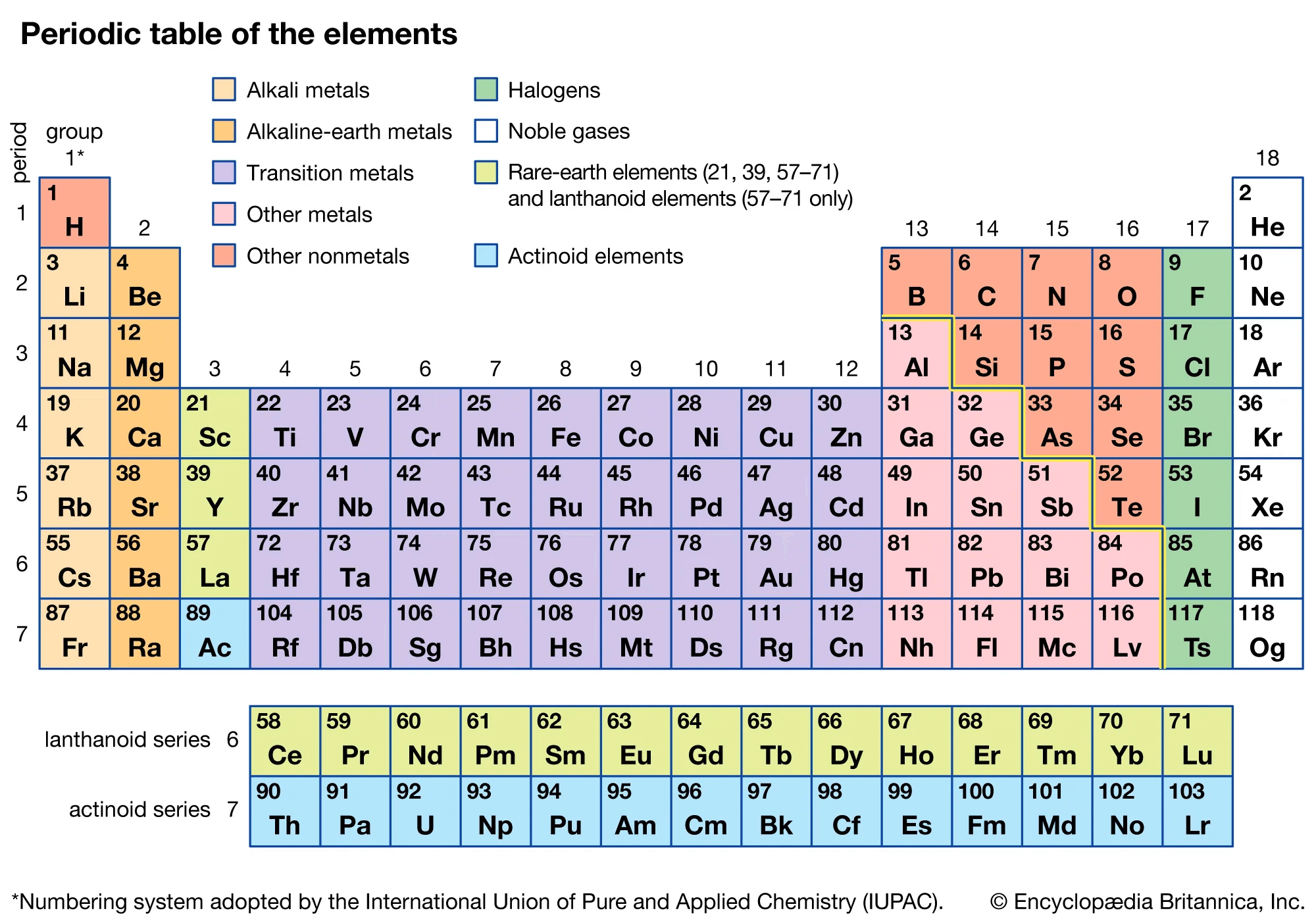
Structure of the Periodic Table
To understand how elements are arranged, it is important to learn about the structure of groups and periods.
Groups vs Periods
| Feature | Groups | Periods |
| Direction | Vertical | Horizontal |
| Number | 18 | 7 |
| Similarity | Same properties | Gradual change in properties |
Elements in the same group have similar chemical properties, while elements across a period show gradual variation.
Blocks of the Periodic Table
Another important way to classify elements is based on their electron configuration, which divides the table into blocks.
| Block | Groups | Characteristics |
| s-block | 1–2 | Highly reactive metals |
| p-block | 13–18 | Metals, non-metals, metalloids |
| d-block | 3–12 | Transition metals |
| f-block | Inner transition | Rare earth elements |
This classification is very important for understanding chemical behavior.
s-Block Elements
Let us now study each block in detail, starting with the s-block elements.
| Feature | Details |
| Groups | 1 & 2 |
| Group 1 | Alkali Metals (Na, K) |
| Group 2 | Alkaline Earth Metals (Mg, Ca) |
| Nature | Highly reactive |
These elements tend to lose electrons easily and are therefore highly reactive metals.
p-Block Elements
Next, we move to the p-block, which contains a wide variety of elements.
| Feature | Details |
| Groups | 13–18 |
| Group 17 | Halogens |
| Group 18 | Noble Gases |
| Nature | Mix of metals and non-metals |
Noble gases are chemically inert and very stable in nature.
d-Block Elements (Transition Metals)
The d-block elements are important due to their unique chemical properties and frequent appearance in exam questions.
| Feature | Details |
| Groups | 3–12 |
| Type | Metals |
| Special Property | Variable oxidation states |
An important fact to remember is that Mercury is the only metal that exists in liquid state at room temperature.
f-Block Elements
The f-block elements are placed separately at the bottom of the periodic table and have special properties.
| Series | Atomic Numbers | Property |
| Lanthanides | 57–71 | Rare earth metals |
| Actinides | 89–103 | Radioactive |
All actinides are radioactive and are important in nuclear chemistry.
Important Trends in the Periodic Table
To score well in exams, understanding periodic trends is essential as many conceptual questions are based on them.
| Property | Trend |
| Atomic Size | Increases down a group |
| Electronegativity | Decreases down a group |
| Metallic Nature | Increases down a group |
| Ionization Energy | Decreases down a group |
Across a period from left to right, these trends generally show opposite behavior.
Quick Revision Table
Before moving to practice, here is a quick summary for revision.
| Topic | Key Point |
| Periodic Table | Arrangement by atomic number |
| Groups | Vertical columns |
| Periods | Horizontal rows |
| s-block | Groups 1–2 |
| p-block | Groups 13–18 |
| d-block | Transition metals |
| f-block | Lanthanides and Actinides |
Practice Periodic Table based MCQs Live
Q1. How many groups are present in the modern periodic table?
Q2. Which group contains noble gases?
Q3. Which block contains transition elements?
Q4. How many periods are present in the modern periodic table?
Q5. Which group of elements is known as alkali metals?
Q6. Which group of elements is called halogens?
Q7. Which block contains noble gases?
Q8. Which of the following is the basis of the modern periodic table?
Q9. Which element is liquid at room temperature?
Q10. Which block elements are known as transition elements?
Q11. The f-block elements are also known as:
Q12. Which of the following is a noble gas?
Q13. Which property increases down a group in the periodic table?
Q14. Which of the following decreases down a group?
Q15. How many elements are present in the modern periodic table?
Quiz Summary
FAQs
Ans. It is a chart that organizes elements by increasing atomic number.
Ans. Properties of elements are periodic functions of their atomic numbers.
Ans. There are 7 horizontal periods in the modern periodic table.
Ans. The modern periodic table consists of 118 known elements.
Ans. d-block elements are known as transition metals.
- Error Spotting Questions for SSC CGL, Download Free Ebook
- Sentence Improvement Questions for SSC CGL, Attempt Now
- How did AIR 1 use Oliveboard to crack SSC CGL 2026? Mock Data Analysis
- Height and Distance Question Types, Formulas, Short Tricks and Tips
- Delhi Police Exam Shift Timings 2026, Check Exam Timings
- SSC CGL 2025 Tier 1 Question Papers, Shift-Wise PYPs, Download PDFs

Hello, I’m a content writer working at Oliveboard. I focus on creating blogs, articles, and educational content that’s simple, clear, and saves time for readers. I believe in writing that adds real value without overcomplicating things. I also have strong knowledge of banking and government exams, which helps me create content that is both accurate and easy to understand. With experience and consistency, I aim to make preparation smoother for every learner.






